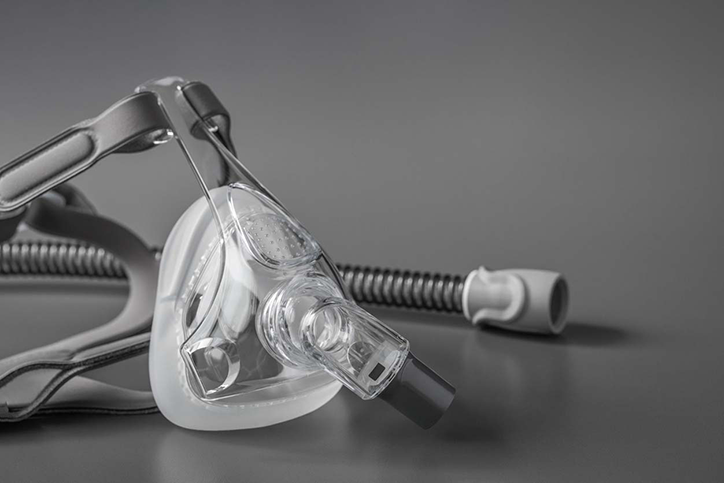
by manovermachine | Aug 18, 2022 | Philips Recalled BiPAP, CPAP, Ventilators
Bilevel Positive Airway Pressure (BiPAP) Machines
The Philips BiPAP device pushes air into your lungs through a mask or nasal plugs that are connected to a ventilator. Patients with chronic obstructive pulmonary disorder (COPD), obstructive sleep apnea, obesity hypoventilation syndrome, pneumonia, asthma flare-up, post-operative breathing difficulties, or neurological disease that disturbs breathing would be candidates for using a BiPAP. Patients with very poor breathing, reduced consciousness, or problems swallowing might not be helped by using a BiPAP.
Continuous Positive Airway Pressure (CPAP) Machines
Philips CPAP devices use mild air pressure from a small, sophisticated air compressor to keep airways open while sleeping. Patients with sleep-related breathing disorders are commonly treated with CPAP machines.
Ventilators
A Philips ventilator is a machine that moves air in and out of the lungs, doing the breathing work that the diaphragm and other muscles normally perform. Ventilators are for patients who are no longer able to breathe normally on their own.
Philips Respironics (Philips) is the maker of multiple types of breathing devices and supplies including BiPAP machines, CPAP machines, and ventilators. In June of 2021, Philips recalled several specific models of these devices, the majority of which are first-generation DreamStation products sold prior to April 2021. This recall involves millions of devices that may be affected by a defect that can harm the user.
These devices use a polyester-based polyurethane (PE-PUR) to lessen sound and vibration. This PE-PUR material can break down and pieces of black foam or certain chemicals could be breathed in or swallowed, potentially resulting in serious injury. The breakdown of this PE-PUR foam may be caused by hot and humid conditions or by the use of ozone cleaners or other cleaning methods not recommended by the manufacturer.
Injuries Due to the Recalled Philips Devices
Respiratory:
- Lung damage
- New or worsening asthma
- Pneumonia
- Respiratory failure (such as Acute Respiratory Distress Syndrome (ARDS))
- Pleural effusion
- Reactive Airway Disease (RAD)
Cancer:
- Blood, Lymph Node, and Oral Cancers:
- Acute Myeloid Leukemia (AML)
- Blood Cancer
- Bone Marrow Cancer
- Esophageal Cancer
- Hematopoietic Cancer
- Laryngeal Cancer
- Leukemia
- Lymphoma
- Multiple Myeloma
- Nasal Cancer
- Non-Hodgkin’s Lymphoma
- Soft Palate Cancer
- Sinus Cancer
- Throat Cancer
- Tonsil Cancer
- Thyroid Cancers:
- Thyroid Cancer
- Papillary Cancer
- Other Cancers:
- Kidney Cancer
- Liver Cancer
- Lung Cancer
- Bladder Cancer
Other:
- Severe ear, eye, nose, throat, sinus, oral cavity inflammation and injury including nodules, cysts, and tumors
- Sarcoidosis (particularly of the lungs and/or lymph nodes) that required treatment
- Kidney damage (acute kidney injury or chronic kidney disease)
- Liver damage (acute liver failure or chronic liver disease)
FDA Recommendations
The FDA has issued recommendations for patients and their caregivers who use recalled ventilators at home, and recommendations for health care providers and facilities. Separate recommendations were issued to patients and their caregivers who use the recalled BiPAP or recalled CPAP devices.
See the list of recalled devices.
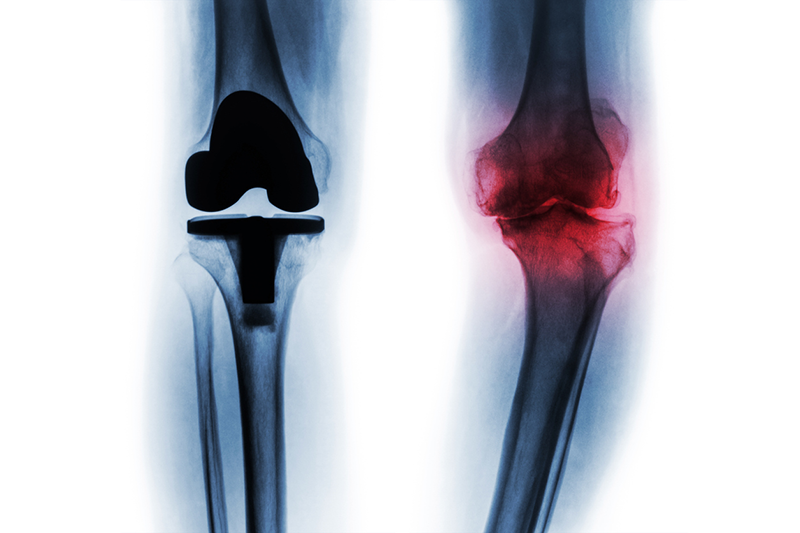
by manovermachine | Aug 18, 2022 | Defective Joint Replacements
Joint replacement surgery, whether it is for a hip, knee, or ankle, is usually necessary when the joint is worn or damaged, resulting in loss of mobility and chronic pain, even when resting. Other less invasive treatments are usually tried before resorting to joint replacement. Conditions that can cause joint damage include osteoarthritis, rheumatoid arthritis, or osteonecrosis. Athletic, accidental, or occupational injuries are common precursors to needing joint replacement.
When pain persists, mobility is affected, sleep is interrupted, and everyday activities become difficult or impossible, replacing a joint can be a huge factor in improving quality of life. As a result, orthopedic surgeons perform more than 450,000 hip replacements each year in the United States, almost 800,000 knee replacements, and only several hundred total ankle replacements.
Signs of a Defective Hip Implant, Knee Implant, or Ankle Implant
- New or worsening pain
- Swelling
- Dislocation
- Inability to bear weight
- Instability
- Clicking, grinding, popping, or other noises
- Osteolysis, or bone degeneration
- Premature wear or device failure
- Revision surgery
Any of these outcomes following joint replacement is cause for concern and when it requires revision surgery, it poses additional risk to the patient.
Exactech Recalls Knee and Ankle Inserts and Liners
Joint replacement products manufacturer Exactech has recalled their polyethylene inserts for knee and ankle replacements because of possible degradation of the polymer construction. Since 1994 hundreds of thousands of their knee and ankle inserts or liners have been implanted and could be involved in the recalls.
Multiple Hip Replacement Recalls
Makers of hip replacement devices have become embroiled in lawsuits filed by people who have suffered severe complications from their devices. Exactech has phased out their Connexion GXL hip liner and will no longer be manufacturing it in the United States. Smith & Nephew has recalled multiple models of their hip systems due to patients who have suffered serious complications requiring revision surgery. Manufacturer Stryker has had multiple implants recalled due to unexpected complications. And Zimmer Biomet, the largest hip implant manufacturer in the U.S. recalled multiple devices that experienced failure.
These manufacturers have faced multiple lawsuits as a result of the recalled products.
by manovermachine | Aug 18, 2022 | Zantac Linked Cancers
According to the National Institutes of Health, the heartburn preventative Zantac was approved for use in the United States in 1983 and by 1988 it had become the world’s best-selling drug. In 2004 generic ranitidine was approved for over-the-counter use in the U.S. and remained available until April 2020, when the FDA announced the withdrawal from the market of prescription, over-the-counter, and generic ranitidine and issued a warning to consumers. Nearly 15 million people were prescribed Zantac yearly prior to it being withdrawn from the market.
The active ingredient in Zantac is a substance called ranitidine and it works by decreasing the amount of acid your stomach makes. In 2018 ranitidine was found to have low levels of the known human carcinogen NDMA. N-nitrosodimethylamine is a stabilizing agent used in gasoline, rocket fuel, and other petroleum-based industrial products. The FDA found that the level of NDMA in ranitidine increased when the drug was stored in high temperatures and also as it got older.
Negative Effects of Zantac Use
According to the FDA, since 1983 there have been 73,240 cases reported with a negative effect from Zantac. This includes 55,891 serious cases, 4,926 deaths, and 66 percent of the cases were linked to cancer. In 2020 alone, however, reports of adverse reactions to Zantac spiked to 71,861. 70 percent of these reports mentioned cancers, the most common one being colorectal or colon cancer.
While the short-term risk of NDMA-caused cancer is thought to be low, there have not been full evaluations of longer-term exposure. However, Zantac and other ranitidine-based drugs have been linked to colorectal, kidney, bladder, prostate, esophageal, stomach, liver, pancreatic, lung, breast, gastrointestinal, uterine, testis, and thyroid cancers. Of the cancer deaths that were tied to taking Zantac, pancreatic cancer had the highest number of deaths, followed by liver, esophageal, colorectal, and stomach cancers.
Demographics of Zantac Injury
According to the FDA Adverse Event Reporting System (FAERS), 12,982 of the cases were reported by females, 8,437 cases were reported by males, but the majority of cases (51,821) did not specify gender.
Age was not specified in 39,002 cases. In the cases that did specify age, there were 19,027 reported cases in the 18-64 year range, 13,168 cases reported in the 65-85 year range, and all other age ranges had fewer than 1,000 cases reported.
Zantac-related cancers can take years to develop, so the serious adverse effects are not known in many cases for around four years.

by manovermachine | Aug 18, 2022 | Ovarian Cancer and Talcum Powder
Located at the ends of the fallopian tubes in the female reproductive system, ovaries are almond-sized glands that produce eggs and in which the female hormones estrogen and progesterone are made. The Mayo Clinic explains that ovarian cancer is a type of cancer that begins in the ovaries, but can multiply quickly and invade and destroy healthy body tissue anywhere in the body.
Ovarian cancer begins when cells in or near the ovaries develop mutations in their DNA. The cancer cells grow and multiply quickly, forming tumors which can break off to spread or metastasize to other parts of the body. Depending on where the cancer begins, determines the type of ovarian cancer that develops and what the best treatment options are.
- Epithelial ovarian cancer is the most common
- Stromal tumors which are rare are usually diagnosed earlier than other ovarian cancers
- Germ cell tumors are rare ovarian cancers that tend to occur at a younger age
Risk Factors for Ovarian Cancer
- Risk increases with age and is most often diagnosed in older women
- Certain gene changes inherited from your parents
- Family history of ovarian cancer
- Being overweight or obese
- Postmenopausal hormone replacement therapy
- Endometriosis
- Early menstruation and/or late menopause
- Never having been pregnant
Taking birth control pills can help reduce your risk of developing ovarian cancer. Genetic testing, especially if there is a family history of breast or ovarian cancers, can help decide whether you should consider surgery to remove your ovaries in order to prevent cancer.
Connection Between Ovarian Cancer and Talcum Powder Use
While Johnson & Johnson’s baby powder is probably the most recognizable brand of talcum powder used by women, there are multiple companies being sued by women diagnosed with ovarian cancer. The cancer-causing ingredient in talcum powder is asbestos, and when it is used on the genital area or on undergarments, sanitary pads, diaphragms or condoms it is thought that it can enter the body and travel through the vagina, uterus, and fallopian tubes to the ovaries. Among the many studies conducted so far on the possible link between talcum powder and ovarian cancer, results have been mixed. Some studies have reported a slightly increased risk and some studies have reported no increase.
Studies have suggested that there is an increased risk of lung cancer and other respiratory diseases among talc miners and millers, due to the asbestos content naturally found in talc. Purified talc in consumer products does not contain asbestos.

by manovermachine | Aug 18, 2022 | Non-Hodgkin's Lymphoma and Roundup
The Mayo Clinic defines Non-Hodgkin’s lymphoma as “a type of cancer that begins in your lymphatic system, which is part of the body’s germ-fighting immune system. In non-Hodgkin’s lymphoma, white blood cells called lymphocytes grow abnormally and can form growths (tumors) throughout the body.”
A blood cancer, there are more than 90 types of non-Hodgkin’s lymphoma (NHL) and approximately 82,000 people are diagnosed with NHL each year in the United States. Any lymphoma that is not chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) or does not involve the Reed-Sternberg cells is classified as a non-Hodgkin’s lymphoma.
To differentiate, Hodgkin’s lymphoma(HL) involves the Reed-Sternberg cells, and there are five types of HL. Reed-Sternberg cells are large, abnormal lymphocytes, which is a type of white blood cell, that may contain more than one nucleus.
Non-Hodgkin’s lymphoma commonly begins in the body’s B cells or T cells, and determining where your NHL originates helps determine treatment options.
Symptoms of NHL
- Swollen lymph nodes
- Fever
- Unexplained weight loss
- Excessive sweating
- Chills
- Lack of energy
- Itching
Since these are common symptoms of many other illnesses, and can often be mistaken as the flu, it’s important to be examined by a doctor in order to rule out lymphoma as the cause.
Risk Factors for NHL
- Immune-suppressing medications, such as those taken after an organ transplant
- Certain viral and bacterial infections, like HIV and Epstein-Barr.
- Certain chemicals used to kill insects and weeds, such as Roundup
- Risk increases with age
Glyphosate is the active ingredient in the weedkiller Roundup that is thought to be carcinogenic. The EPA is still reviewing data on the herbicide to determine whether it is harmful or not, however courts have taken up Roundup lawsuits and have been awarding damages to people who blame Roundup for their non-Hodgkin’s lymphoma. Roundup is still available in 130 countries and is approved for 100 different kinds of crops.
In 1996, Monsanto began marketing “Roundup Ready” seeds for various crops that were genetically modified to be immune to glyphosate. As a result, glyphosate use by farmers increased as much as triple what they were using. By 2016, glyphosate use increased nearly 15-fold. A 2019 study in the journal Mutation Research/Reviews in Mutation Research reported a link between long-term, high-use exposure to glyphosate and a 41 percent increase in the risk of developing non-Hodgkin’s lymphoma.
Lymphoma Research Foundation Resources
Fact Sheets & Guides
Education Programs
LRF Helpline




